Jaclyn R. Lunger, Naomi Lutz, Jiayu Peng, Michal Bajdich, and Yang Shao-Horn
DOI: 10.1021/acs.chemmater.2c00602
Abstract:
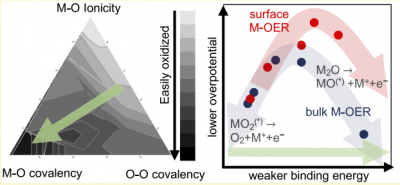
Direct electrolytic extraction of metals from metal oxides is a promising process for the sustainable production of metals. In this work, we elucidate the inherent thermodynamic driving forces behind the reduction of metal oxides to metals (M‑OER). It is shown that the thermodynamics of M‑OER can be systematically tuned via the interactions of oxygen with the participating metal cations as a function of metal–oxygen covalency, oxygen–oxygen covalency, and metal–oxygen ionicity. We screen both group 1 elements and metals that are able to exist in the +2 oxidation state for M‑OER thermodynamics. Li, Fe, and Co are identified as having low thermodynamic overpotentials for electrolytic extraction from their metal oxides due to interactions between oxygen and these metals being neither too strong (covalent) nor too weak (ionic). We further show that the bulk formation energies are predictive of M‑OER reaction energetics on surfaces by developing unified design principles for tuning the thermodynamics of these reduction reactions both in bulk oxides and on surfaces.
