Subhash Chandra, Younggyu Kim, Daniele Vivona, Iradwikanari Waluyo, Adrian Hunt, Christoph Schlueter, Jeong Beom Lee, Yang Shao-Horn and Bilge Yildiz
DOI: 10.1039/D1TA08853J
Abstract:
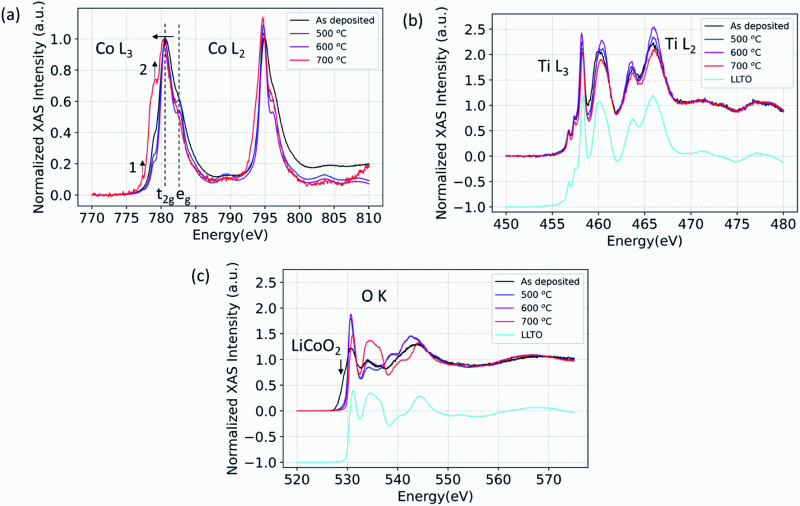
A critical bottleneck for developing successful solid-state batteries is minimizing the interfacial impedances between the solid electrolyte and the active electrode materials. Advancing our understanding of the chemically and electro-chemically induced reactions of the cathode–electrolyte interface is important to achieve stable interfaces with desired charge transfer properties. The perovskite Li-solid electrolyte with the general formula Li3xLa2/3−x□1/3−2xTiO3 (LLTO) is an interesting candidate for solid-state Li-ion cells due to its superionic Li-ion conductivity. However, the chemical compatibility of LLTO with potential cathode materials is not sufficiently understood, and here we examine the LLTO interface with thin-film LiCoO2 (LCO) cathode layers. We show that elemental mixing, in particular Co diffusion into LLTO, and increased structural ordering of the LCO occurs at moderate temperatures, on-setting around 300 °C in air, even in the absence of any secondary phases. This reaction decreases the LCO|LLTO interface charge transfer resistance by almost 50% after annealing to 500 °C relative to the as-deposited state. Reduction of the interface charge transfer resistance can be attributed to the formation of a mixed electronic and ionic conducting zone near the interface upon Co diffusion into LLTO. This finding can guide the engineering of cathode–solid electrolyte interfaces with fast charge transfer kinetics.
